
The periodic table below is color coded to show. The elements that touch the stair-step line are the metalloids. Semi metals exhibit properties intermediate between those of metals and nonmetals. This line divides the metals from the nonmetals. 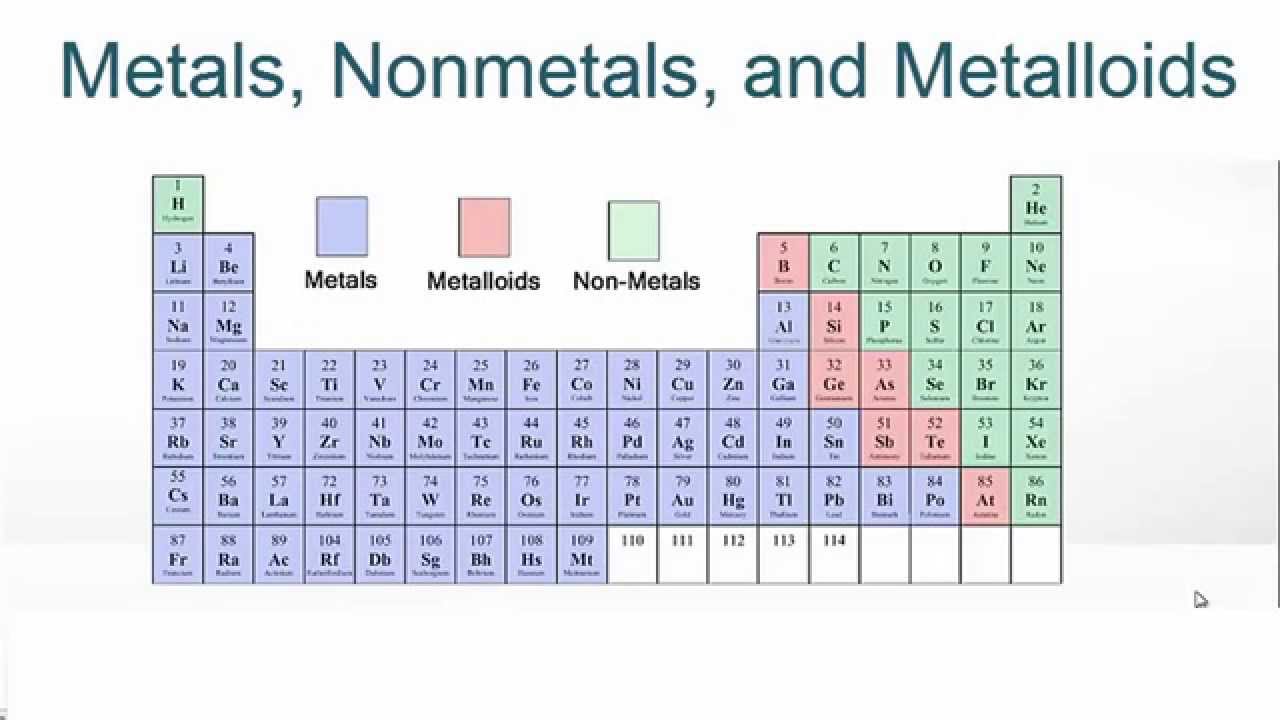
Also, many periodic tables have a stair-step line on the table identifying the element groups. The metalloids separate the metals and nonmetals on a periodic table. Distinguishing luster (shine) Non-lustrous, various colors. The elements can be broadly divided into metals, nonmetals, and semi metals. Updated on JanuElements of the periodic table are grouped as metals, metalloids or semimetals, and nonmetals. Table 2.11.1: Characteristic properties of metallic and non-metallic elements: Metallic Elements. Some of the groups have widely-used common names, including the alkali metals (Group 1) and the alkaline earth metals (Group 2) on the far left, and the halogens (Group 17) and the noble gases (Group 18) on the far right. Elements that exhibit similar chemistry appear in vertical columns called groups (numbered 1–18 from left to right) the seven horizontal rows are called periods. The periodic table is an arrangement of the elements in order of increasing atomic number. Each group is located in a different part of the periodic table. For example, an electronegativity periodic table color codes the elements based on how electronegative they are. The noble gases are the group of nonmetals found on the righthand side of the periodic table. 
So, the least metallic elements are the noble gases even though some people forget they are a subset of the nonmetals. Periodic tables can use color to identify other element characteristics. Metallic character decreases as you move from left to right across the periodic table. Color coding helps identify similar elements such as these at a glance. Elements are either metals, nonmetals, or metalloids (or semi metals). The metalloids and nonmetals, for example, don't fall neatly into the same column.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
